I'm often sent tips by followers and friends of my blog. This one was of particular interest to me, sent to me by a fellow Facebooker, who shall be known as "Susan."
The reason? Well, it's kinda difficult to read between the lines because, as usual, any funding aimed in the direction of children's well-being is vague where pharmaceutical companies are concerned.
A
press release from Education North Carolina (EDNC) shows us that GlaxoSmithKline and CISNC have
"joined together to announce tools and training to ensure that students in the state’s lowest performing schools have access to high quality wraparound services to propel success in school and beyond."
Tools and training?
The press release goes on to say that...
Yesterday at CISNC’s “2015 Impact Training: Mission Possible” event in Cary, GSK announced a $360,000 grant to enable Communities In Schools of North Carolina to put high quality, research-based interventions in schools to increase attendance, improve behavior, enhance coursework, and engage more parents and families.
Should we be concerned about this or should we be applauding Glaxo?
Exactly what are research-based interventions that will improve behavior? Sadly, the press release doesn't elaborate.
I did some digging.
Glaxo, who have a HQ in Research Triangle Park, N.C, seem to be caring a lot about their community in North Carolina. On the surface, this looks good for GSK.
In 2013, GlaxoSmithKline and Community Care of North Carolina (CCNC) announced the initiation of a new approach to analyzing health information that allowed healthcare providers and payers, with the touch of a button, to analyze patients' medication challenges in real time to determine "effective ways to overcome barriers to delivering quality care."
The service utilizes "prescriptive analytics" to analyze the data and determine suggested interventions that are most likely to facilitate better patient engagement and ultimately, improved patient outcomes.
They added...
... the goal of the project is to assist these healthcare professionals in determining – and responding to medication-related problems that often lead patients to use their medicine incorrectly, or to not adhere to the treatment regimen prescribed by their doctor.
Hmm.
So, I'll ask again, exactly what are research-based interventions that will improve behavior?
A quick glance of CISNC's page
shows that
Lisa Benna of GlaxoSmithKline is a board member. Benna is
Human resources vice president at Glaxo.Previously she worked for Wyeth in a range of roles in both organization development and HR roles in R&D, Manufacturing and Commercial.
Eric Hall (Pictured above), who is the president and CEO of the Communities In Schools of North Carolina, was previously National Director of Educational Services where, according to his
LinkedIn page, he managed and supported a network of over 50 non-traditional schools for students at-risk of dropout across nine states. In his role, he worked to establish value-added models of teacher evaluation, implement common core standards, develop teacher and school leader professional development pathways, and also worked closely with state and local policy-makers to ensure effective practices for serving at-risk students.
I understand that there are problem students, what I can't quite grasp here is why CISNC would team up with a pharmaceutical company who has a pretty poor record when it comes to the safety of children. Is it just purely about the $360,000 grant GSK have given them?
Will they be influenced by GSK's kind offering, will those children with behavoral problems, who, after being offered one-to-one adult supervision, be then sent down the mental health road and prescribed GlaxoSmithKline drugs such as Wellbutrin, Paxil, Lamictal, all medicines used to treat mood disorders?
Maybe I'm clutching at straws here. Maybe GlaxoSmithKline have turned a corner and are trying to make amends for their previous abhorrent treatment of children with Paxil. Maybe it's just a huge PR exercise or maybe Glaxo have ulterior motives?
Whatever way you slice it, GlaxoSmithKline have pleaded guilty for promoting medicines to kids, medicines that were unsafe for kids. Why would any organisation connected with children wish to have GSK on board?
It's a head scratcher for me.
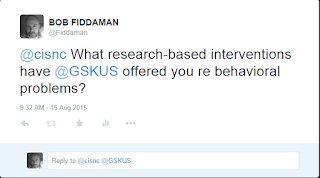
I Tweeted the following to CISNC - they never replied.
So, I fired off an email to Jill Cox, who is the Communities In Schools of North Carolina contact. I basically want to know what these research-based interventions are and whether or not they involve the use of psychiatric medication.
Dear Jill,
I read with great interest that CISNC have recently joined forces with GlaxoSmithKline and announced tools and training to ensure that students in lowest performing schools have access to high quality wraparound services to propel success in school and beyond.
The press release, I read, also states that "Yesterday at CISNC’s “2015 Impact Training: Mission Possible” event in Cary, GSK announced a $360,000 grant to enable Communities In Schools of North Carolina to put high quality, research-based interventions in schools to increase attendance, improve behavior, enhance coursework, and engage more parents and families."
Exactly what are research-based interventions that will improve behavior?
I'd be grateful if you could let me know the answer to this at your earliest convenience.
Sincerely,
--
Bob Fiddaman
I will, as usual, keep you updated if Jill responds.
Now, here's something that I bet you all thought you'd never read, from me at least.
I think it's safe to say that I am probably one of Glaxo's biggest online critics but I have to take my hat off to them and, probably for the first time ever, give them praise, particularly for looking after the residents of North Carolina. Credit where credit is due, I guess.
The
North Carolina GlaxoSmithKline Foundation offers grants to various organisations and charities across the state of NC. Since 1986 they have awarded $65 million in grants to North Carolina nonprofit organizations. I do applaud them for that. However, this should not, nae should never, detract from the fact that GlaxoSmithKline have caused a lot of harm to a lot of people with their antidepressant Paxil - there are other prescription medications manufactured by Glaxo that have also caused death and heartache, Avandia for one.
When we balance up the good that they have, it appears, done for these nonprofit organizations in North Carolina against the bad they have done with drugs such as Paxil and Avandia we still see a company that is almost Jekyll and Hyde-like. It's almost as if grants such as the above are handed out to ease the conscience of their abhorrent history. It would be far better, in my opinion, if GlaxoSmithKline were seen to hand out cash awards to those they have harmed and not make them, or their families, have to jump through legal hoops in Paxil suicide litigation, Paxil withdrawal litigation and Paxil birth defect litigation. Glaxo will only ever settle when they know they cannot win trials. It seems absurd, therefore, that they seem to paint themselves whiter than white when handing out cash awards to nonprofit organizations across North Carolina. For families affected by Paxil, be they families who have suffered a loss of a child or adult due to Paxil induced suicide or mothers who have given birth to children with heart defects due to Paxil ingestion during pregnancy or for those affected by the horrendous withdrawal problems Paxil has caused them, seeing Glaxo hand out $65 million to organizations must be a kick in the teeth.
On one hand they giveth, on the other hand their drug, Paxil, taketh away.
And that just doesn't sit right with me. It never will.
Bob Fiddaman.